
Are you researching drug labels, the regulatory intelligence landscape, scientific literature and clinical trials with multiple open browser tabs? Are you finding and comparing multiple sources, piecing together an answer, and manually tracking everything on spreadsheets? There's a better way.
Trust Dr.Evidence, the first and most experienced innovator to bring life sciences intelligence into one location to streamline your work. We bring pharmaceutical labeling, regulatory, commercial, safety, clinical trials and scientific literature into a single, unified platform. Transform how your team works— from regulatory submission through commercialization— with a platform that accelerates decision-making, de-risks strategy, and delivers results you can trust.
time to insight, conducting complex regulatory analysis in hours instead of weeks.

labels, drug applications, clinical trials and scientific literature into a unified workflow.

fragmented evidence from multiple sources into a clear decision pathway.

time-intensive and manual tasks so you can focus on strategic work at the top of your capabilities.

securely in-platform with colleagues across your organization.

your analysis with human-in-command fit-for-purpose AI summaries and insights.

Created the pharma landscape intelligence space, with continuous innovation

20+ years of domain experience

25+ proprietary AI models with zero IP leakage

Complimentary white-glove search optimization and ongoing support

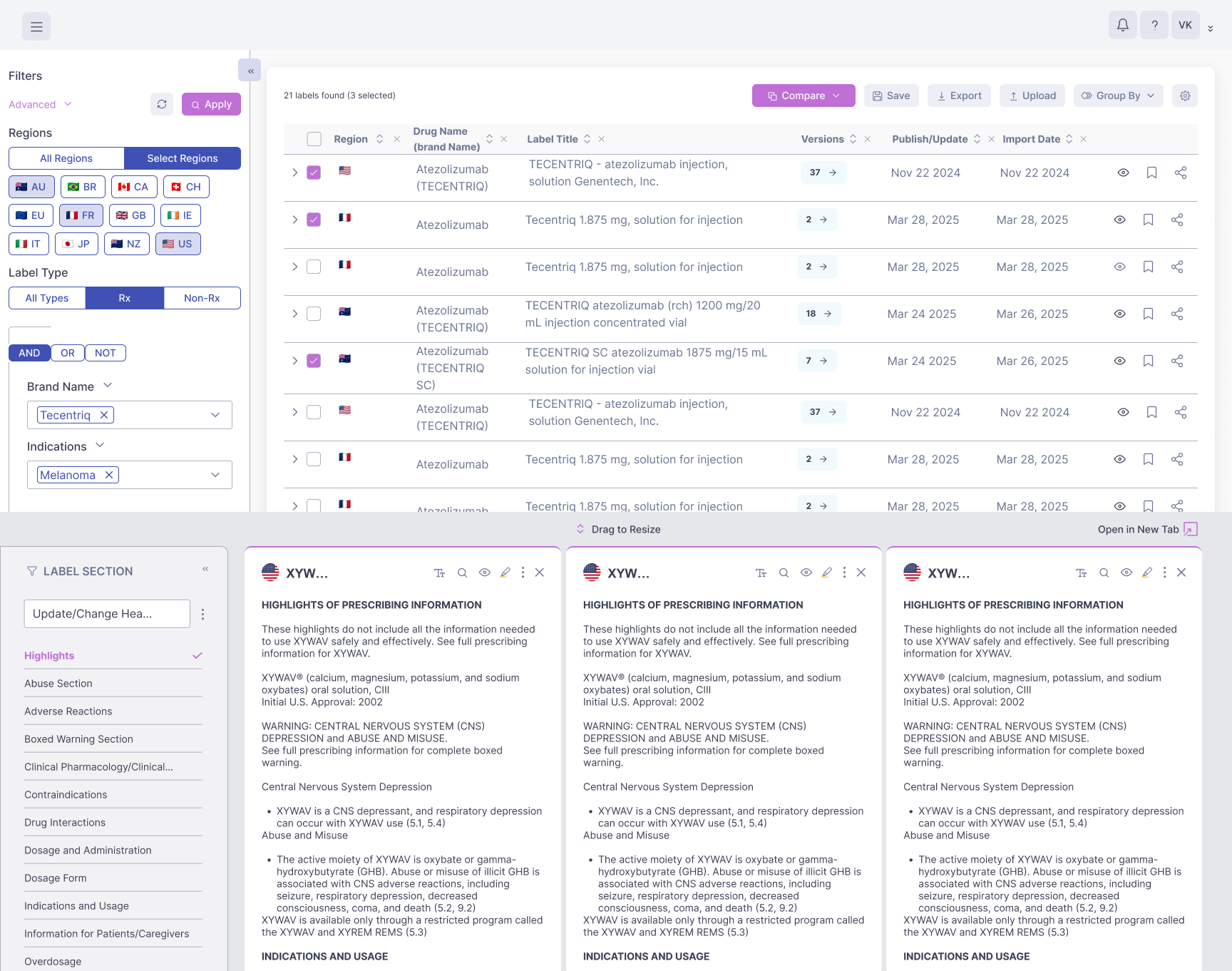
728,000+ labels from 11 major health authorities
Centralizes U.S. drug labels from Drugs@FDA, DailyMed, Orange Book, Purple Book, and more
Generate side-by-side comparisons instantly
![[background image] image of dispatch center (for a courier & delivery service)](https://cdn.prod.website-files.com/69ab1113a9b4602c0bd3716c/69b042e106e16887586a5bb6_Structured%20Search%20-%20Trials%20Intelligence%20-%20Search%20Results%203.png)
1.1 million clinical trials
70 million scientific literature citations
400,000 congresses
![[background image] image of dispatch center (for a courier & delivery service)](https://cdn.prod.website-files.com/69ab1113a9b4602c0bd3716c/69b044c7ae74156f33533b08_Structured%20Search%20-%20Lit%20Intelligence%20-%20Search%20Results.png)
31 million patents and 500,000 NIH grants
2.1 million articles from healthcare RSS feeds
![[background image] image of dispatch center (for a courier & delivery service)](https://cdn.prod.website-files.com/69ab1113a9b4602c0bd3716c/69b045f9559cb383d82b482a_Structured%20Search%20-%20Reg%20Documents%20-%20Search%20Results.png)
140,000 FDA and EMA approval packages
4,000 FDA and EMA Guidance documents
49,000 Orange and Putple Book data points
Drug labels, regulatory approval packages and guidance published by health authorities across the globe, clinical trials from WHO ICTRP, and scientific literature from PubMed and a range of other reputable sources.

with AI-powered workflows built specifically for biopharma

so your experts can focus on high-value strategy
with contemporary, evidence-driven market insights

by eliminating manual work and automating monitoring

Whether you’re tracking label changes, preparing submissions, or analyzing competitive prior precedent, Dr.Evidence helps you move faster with confidence. Reach out, and our team will follow up to understand your needs and show you what’s possible.

Join the leading biopharma companies worldwide who've made Dr.Evidence their competitive advantage. Get the landscape intelligence platform that grows with you—from first submission to long-term commercial success.

Find quick solutions to common questions about our platform and services.
Dr.Evidence is the AI-powered labeling and regulatory landscape intelligence platform designed specifically for biopharma companies. As an Authoritative Source, we help labeling and regulatory professionals accelerate time-to-market by providing access to more than 100 million documents. This includes:
· Drug labels
· Regulatory approval packages and guidance documents
· Clinical trials
· Scientific literature
These combined with 25+ proprietary AI models, deliver actionable insights throughout the drug development lifecycle.
Dr.Evidence automates routine research tasks and delivers evidence-based insights in minutes instead of weeks. Our platform uses generative AI, machine learning, and natural language processing to help you:
· Rapidly analyze global regulatory landscapes
· Identify relevant precedents
· Compare competitor labels
· Extract critical information from approval packages.
This lets your labeling and regulatory teams to focus on strategic decision-making instead of manually reviewing documents, significantly reducing time-to-market.
It provides access to 100 million+ documents from health authorities worldwide, including:
Drug labels and product information from FDA, EMA, PMDA, Health Canada, TGA, and more
Regulatory approval packages and guidance documents
Clinical trial data from WHO ICTRP
Scientific literature from PubMed and other reputable sources
Yes, security and IP protection are fundamental to our platform. Dr.Evidence guarantees zero IP leakage—your proprietary data never trains our models or becomes accessible to other users. We maintain enterprise-grade security protocols and compliance standards required by top-tier pharmaceutical companies. Our 20+ years of biopharma domain expertise means we understand the importance of confidentiality in drug development.
Setup is fast and seamless. Most users are up and running within one business day.
Dr.Evidence is trusted by leading biopharma companies worldwide—from emerging biotechs to top-tier pharmaceutical organizations. Our platform serves labeling and regulatory professionals across branded and generic drug development. Companies choose Dr.Evidence when they need precision, reliability, and comprehensive global regulatory intelligence to maintain their competitive advantage. Our top ten clients have been with us on average for fifteen years.
Unlike general-purpose AI tools, Dr.Evidence is purpose-built for biopharma labeling and regulatory work. Our platform combines:
Authoritative, curated content: Only verified regulatory and scientific sources—no internet noise
Specialized AI models: 25+ proprietary models trained specifically for biopharma workflows
Domain expertise: 20+ years of life sciences knowledge embedded in every feature
Regulatory-grade accuracy: Results you can trust for submissions and strategic decisions
Zero IP risk: Your data stays private and never trains our models
Complimentary optimization: White-glove search support to ensure optimal results
Dr.Evidence leverages three complementary AI approaches optimized for biopharma applications:
Generative AI: Delivers intelligent insights, answers complex questions, and creates comprehensive summaries
Machine learning: Enables precise comparisons, content identification, and automated data extraction
Natural language processing: Provides smart recommendations and accurate language translations
These 25+ proprietary models work together to analyze regulatory landscapes, identify patterns, and surface actionable intelligence that would take weeks to compile manually.
Getting started is simple. Schedule a demo with our team to see how Dr.Evidence can transform your labeling and regulatory workflows. During the demo, we'll show you how our AI-powered platform works with your specific use cases and discuss how we can support your team from first submission through long-term commercial success. Our experts will also create a proprietary library and provide complimentary white-glove search optimization to ensure you get the most robust, reliable results from day one.